window._wpemojiSettings = {"baseUrl":"https:\/\/s.w.org\/images\/core\/emoji\/15.0.3\/72x72\/","ext":".png","svgUrl":"https:\/\/s.w.org\/images\/core\/emoji\/15.0.3\/svg\/","svgExt":".svg","source":{"concatemoji":"https:\/\/www.uniquenewsonline.com\/wp-includes\/js\/wp-emoji-release.min.js?ver=6.5.2"}};
/*! This file is auto-generated */
!function(i,n){var o,s,e;function c(e){try{var t={supportTests:e,timestamp:(new Date).valueOf()};sessionStorage.setItem(o,JSON.stringify(t))}catch(e){}}function p(e,t,n){e.clearRect(0,0,e.canvas.width,e.canvas.height),e.fillText(t,0,0);var t=new Uint32Array(e.getImageData(0,0,e.canvas.width,e.canvas.height).data),r=(e.clearRect(0,0,e.canvas.width,e.canvas.height),e.fillText(n,0,0),new Uint32Array(e.getImageData(0,0,e.canvas.width,e.canvas.height).data));return t.every(function(e,t){return e===r[t]})}function u(e,t,n){switch(t){case"flag":return n(e,"\ud83c\udff3\ufe0f\u200d\u26a7\ufe0f","\ud83c\udff3\ufe0f\u200b\u26a7\ufe0f")?!1:!n(e,"\ud83c\uddfa\ud83c\uddf3","\ud83c\uddfa\u200b\ud83c\uddf3")&&!n(e,"\ud83c\udff4\udb40\udc67\udb40\udc62\udb40\udc65\udb40\udc6e\udb40\udc67\udb40\udc7f","\ud83c\udff4\u200b\udb40\udc67\u200b\udb40\udc62\u200b\udb40\udc65\u200b\udb40\udc6e\u200b\udb40\udc67\u200b\udb40\udc7f");case"emoji":return!n(e,"\ud83d\udc26\u200d\u2b1b","\ud83d\udc26\u200b\u2b1b")}return!1}function f(e,t,n){var r="undefined"!=typeof WorkerGlobalScope&&self instanceof WorkerGlobalScope?new OffscreenCanvas(300,150):i.createElement("canvas"),a=r.getContext("2d",{willReadFrequently:!0}),o=(a.textBaseline="top",a.font="600 32px Arial",{});return e.forEach(function(e){o[e]=t(a,e,n)}),o}function t(e){var t=i.createElement("script");t.src=e,t.defer=!0,i.head.appendChild(t)}"undefined"!=typeof Promise&&(o="wpEmojiSettingsSupports",s=["flag","emoji"],n.supports={everything:!0,everythingExceptFlag:!0},e=new Promise(function(e){i.addEventListener("DOMContentLoaded",e,{once:!0})}),new Promise(function(t){var n=function(){try{var e=JSON.parse(sessionStorage.getItem(o));if("object"==typeof e&&"number"==typeof e.timestamp&&(new Date).valueOf()<e.timestamp+604800&&"object"==typeof e.supportTests)return e.supportTests}catch(e){}return null}();if(!n){if("undefined"!=typeof Worker&&"undefined"!=typeof OffscreenCanvas&&"undefined"!=typeof URL&&URL.createObjectURL&&"undefined"!=typeof Blob)try{var e="postMessage("+f.toString()+"("+[JSON.stringify(s),u.toString(),p.toString()].join(",")+"));",r=new Blob([e],{type:"text/javascript"}),a=new Worker(URL.createObjectURL(r),{name:"wpTestEmojiSupports"});return void(a.onmessage=function(e){c(n=e.data),a.terminate(),t(n)})}catch(e){}c(n=f(s,u,p))}t(n)}).then(function(e){for(var t in e)n.supports[t]=e[t],n.supports.everything=n.supports.everything&&n.supports[t],"flag"!==t&&(n.supports.everythingExceptFlag=n.supports.everythingExceptFlag&&n.supports[t]);n.supports.everythingExceptFlag=n.supports.everythingExceptFlag&&!n.supports.flag,n.DOMReady=!1,n.readyCallback=function(){n.DOMReady=!0}}).then(function(){return e}).then(function(){var e;n.supports.everything||(n.readyCallback(),(e=n.source||{}).concatemoji?t(e.concatemoji):e.wpemoji&&e.twemoji&&(t(e.twemoji),t(e.wpemoji)))}))}((window,document),window._wpemojiSettings);
https://www.uniquenewsonline.com/wp-includes/js/jquery/jquery.min.js
https://www.uniquenewsonline.com/wp-includes/js/jquery/jquery-migrate.min.js
var pbLocalizeObj = {"ajax":"https:\/\/www.uniquenewsonline.com\/wp-admin\/admin-ajax.php","seconds":"seconds","thisWillClose":"This will close in","icons":{"close_icon":"<svg class=\"ays_pb_material_close_icon\" xmlns=\"https:\/\/www.w3.org\/2000\/svg\" height=\"36px\" viewBox=\"0 0 24 24\" width=\"36px\" fill=\"#000000\" alt=\"Pop-up Close\"><path d=\"M0 0h24v24H0z\" fill=\"none\"\/><path d=\"M19 6.41L17.59 5 12 10.59 6.41 5 5 6.41 10.59 12 5 17.59 6.41 19 12 13.41 17.59 19 19 17.59 13.41 12z\"\/><\/svg>","close_circle_icon":"<svg class=\"ays_pb_material_close_circle_icon\" xmlns=\"https:\/\/www.w3.org\/2000\/svg\" height=\"24\" viewBox=\"0 0 24 24\" width=\"36\" alt=\"Pop-up Close\"><path d=\"M0 0h24v24H0z\" fill=\"none\"\/><path d=\"M12 2C6.47 2 2 6.47 2 12s4.47 10 10 10 10-4.47 10-10S17.53 2 12 2zm5 13.59L15.59 17 12 13.41 8.41 17 7 15.59 10.59 12 7 8.41 8.41 7 12 10.59 15.59 7 17 8.41 13.41 12 17 15.59z\"\/><\/svg>","volume_up_icon":"<svg class=\"ays_pb_fa_volume\" xmlns=\"https:\/\/www.w3.org\/2000\/svg\" height=\"24\" viewBox=\"0 0 24 24\" width=\"36\"><path d=\"M0 0h24v24H0z\" fill=\"none\"\/><path d=\"M3 9v6h4l5 5V4L7 9H3zm13.5 3c0-1.77-1.02-3.29-2.5-4.03v8.05c1.48-.73 2.5-2.25 2.5-4.02zM14 3.23v2.06c2.89.86 5 3.54 5 6.71s-2.11 5.85-5 6.71v2.06c4.01-.91 7-4.49 7-8.77s-2.99-7.86-7-8.77z\"\/><\/svg>","volume_mute_icon":"<svg xmlns=\"https:\/\/www.w3.org\/2000\/svg\" height=\"24\" viewBox=\"0 0 24 24\" width=\"24\"><path d=\"M0 0h24v24H0z\" fill=\"none\"\/><path d=\"M7 9v6h4l5 5V4l-5 5H7z\"\/><\/svg>"}};
https://www.uniquenewsonline.com/wp-content/plugins/ays-popup-box/public/js/ays-pb-public.js
var breeze_prefetch = {"local_url":"https:\/\/www.uniquenewsonline.com","ignore_remote_prefetch":"1","ignore_list":["\/wp-admin\/"]};
https://www.uniquenewsonline.com/wp-content/plugins/breeze/assets/js/js-front-end/breeze-prefetch-links.min.js
Unique News Online
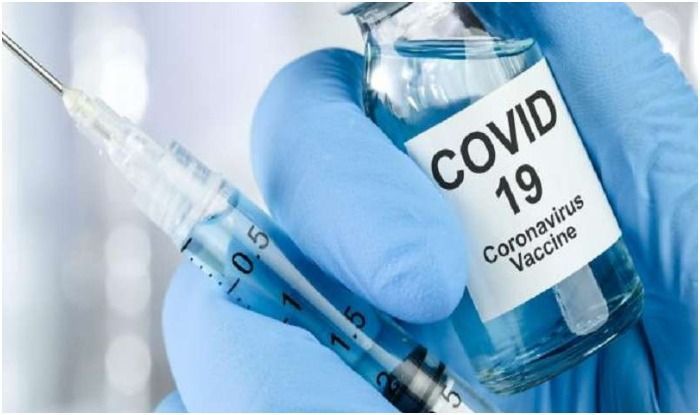
Indian Health Experts Take Russian COVID-19 Vaccine With Pinch of Salt
Indian Health Experts Take Russian COVID-19 Vaccine With Pinch of Salt Published
4 years ago on
August 12, 2020
Cautiously welcoming the information of Russia registering the world’s first Covid-19 vaccine, Indian well being consultants on Tuesday harassed that it will be too early to say whether it is protected or not because the vaccine was examined on only a few volunteers. Additionally Learn – Will Critically Analyse Its Impact: AIIMS Director Randeep Guleria on Russia’s COVID-19 Vaccine
The medical trials of the vaccine began on June 18 and included 38 volunteers. Additionally Learn – Pretend Information: The Lady Receiving Covid-19 Vaccine in This Viral Video is Not Vladimir Putin’s Daughter!
The volunteers had been divided into two essential teams – one group consisted of 18 folks and the opposite group had 20.
Russia on Tuesday grew to become the primary nation on the planet to register a Covid-19 vaccine collectively developed by the Gamaleya Analysis Institute and the Russian Defence Ministry.
In response to Dr Rajesh Chawla, Senior Advisor, Respiratory and Crucial Care at Indraprastha Apollo Hospitals in New Delhi, little or no is thought about this vaccine because it claims to assist folks construct immunity towards this novel virus.
“It is rather early to measure its efficacy and security,” he informed IANS.
In response to him, loads of parameters like age, folks with a number of co-morbidities, the likelihood of allergic reactions and the likes must be monitored earlier than the vaccine is mass-produced.
Talking to IANS, Dr Praveen Gupta, Director and Head, Neurology at Fortis Memorial Analysis Institute at Gurugram, stated: “The Russian vaccine based mostly on an inanimate protein of adenovirus has been dropped at market with very quick time period trials of only a few volunteers”.
As a medical precept, earlier than any vaccination is used for large-scale public use, “it’s advisable in view of security and enough prevention of a communicable illness that correct and long-duration trials are carried out earlier than the medical use of the vaccine,” the physician harassed.
The World Health Group (WHO) has additionally raised issues concerning the vaccine.
“It’s too early to inform whether it is protected or not. Perhaps in two months, the efficacy of the vaccine might be decided,” stated Gupta.
In response to Dr Jyoti Mutta, Senior Advisor, Microbiology at Sri Balaji Motion Medical Institute in New Delhi, contemplating the pandemic and emergency-like scenario everywhere in the world, the Covid-19 vaccine from Russia has raised hopes.
“However security and efficacy are essential and we have to keep watch over its upcoming outcomes too,” Mutta informed IANS.
“Each vaccine must cross all mandatory trials and levels, and the numerous outcomes of any vaccine are seen in a minimal of one yr with mass customers however contemplating the scenario everybody appears to be in a rush. We ought to be hopeful however sensible as nicely,” she famous.
Passionate news enthusiast with a flair for words. Our Editorial Team author brings you the latest updates, in-depth analysis, and engaging stories. Stay informed with their well-researched articles.
International Workers’ Memorial Day 2024: Current Theme, Quotes, Images, Messages, Posters, Banners, Wishes, Slogans and Captions Published
3 hours ago on
April 26, 2024 International Workers’ Memorial Day is observed on 28 April every year. It’s a day when we remember people who lost their lives at work. According to 2022-23 statistics, around 145 workers lost their lives in the UK due to severe injuries at work.
International Workers’ Memorial Day (IWMD) gives a platform to the trade union movement to highlight the ongoing neglect of the safety and health of workers and to campaign for better protection measures in the work environment.
No one deserves to come home from a workplace with poor health and unsafe practices. There are many workplaces where employers care more about profits than effective work measures.
The day is observed for those who were killed in accidents in the workplace or due to diseases caused by the nature of work. Many organizations around the globe host events and seminars that honor the families of workers who passed away at work.
Some other activities include laying wreaths, multi-faith religious services, raising public awareness of issues, unveiling monuments, and planting trees.
It should be noted that safety and health representatives in the workplace must never be understated. They represent members who ensure employers follow their legal obligations. H&S reps are legally obliged to investigate any mishaps or accidents.
They investigate complaints made by members as well as act as representatives on the employer’s behalf.
It should be noted that transportation causes more deaths in the workplace than in other work environments. The next common reasons for workplace deaths are slips and falls, workplace violence (human and animal), exposure to harmful substances, and contact with poisonous objects and equipment.
International Workers’ Memorial Day 2024 Current Theme This year’s theme will be: “Good Jobs. Safe Jobs. Protect Our Rights.”
Quotes, Images, Messages, Posters On the occasion of Workers’ Memorial Day, let us come together and offer prayers to all those workers who lost their lives while working in the industries.
The occasion of Workers’ Memorial Day reminds us all that we have to join hands in order to make the working conditions safe and better for our workers.
Warm wishes on the occasion of Workers’ Memorial Day to all. We have lost so many workers in all these years in our industries. Let us offer them our prayers.
The safety at workplace is one of the most important concerns for any company. Wishing a very Happy Workers’ Memorial Day.
Each and every worker is important and so is his life. Warm greetings on Workers’ Memorial Day to all. Let us work to make their conditions better.
On International Workers’ Memorial Day, we honor the dedication and sacrifice of workers worldwide. May their contributions never be forgotten.
Remembering the lives lost and the struggles endured for workplace safety and dignity. Wishing everyone a meaningful International Workers’ Memorial Day.
Today, we pay tribute to those who have lost their lives or been injured while on the job. Let’s continue to strive for safer working conditions for all. #IWMD
Sending solidarity and support to workers everywhere on International Workers’ Memorial Day. Your resilience and commitment inspire us all.
As we commemorate International Workers’ Memorial Day, let’s renew our commitment to advocating for the rights and protections of workers worldwide.
Banners, Wishes, Slogans, and Captions May the memories of those who have perished due to workplace accidents or hazards serve as a reminder of the ongoing need for safety measures and worker protections.
On this solemn day, we honor the memory of those who have lost their lives while earning a living. Let’s work together to prevent future tragedies. #IWMD
Reflecting on the sacrifices made by workers across the globe. Wishing peace and strength to their families and colleagues on International Workers’ Memorial Day.
Let’s stand together in solidarity with workers everywhere as we commemorate International Workers’ Memorial Day. Their legacy lives on in our continued fight for justice.
Today, we remember and honor the workers who have lost their lives in pursuit of a better future for themselves and their families. #IWMD
World Intellectual Property Day 2024: Current Theme, Quotes, Images, Messages, Posters, Banners, Cliparts and Instagram Captions Published
18 hours ago on
April 25, 2024 World Intellectual Property Office (WIPO) is observed on 26 April every year. The day is all about promoting the importance of intellectual property (IP) in promoting creativity and innovation.
Many use this day as an opportunity to raise awareness about the requirements of a balanced approach to intellectual property protection as it protects the rights of copyright holders as well as creators in the public interest.
World Intellectual Property Day 2024 Theme Every year a new theme is introduced, this year’s theme will be ‘IP and the SDGs: Building Our Common Future with Innovation and Creativity’.
As mentioned above, the day gives a platform to connect with entrepreneurs, inventors, stakeholders and IP offices to learn and promote IP solutions. It can contribute to our country’s economic development as well as social well-being. Furthermore, it aims to raise the importance of IP protection as well as the role of intellectual property in the field of creativity.
The origin of the day goes back to 1883 when the Paris Convention for the Protection of Industrial Property was officially signed. The convention helped to make intellectual property protections for trademarks, inventions along other industrial designs.
By 1970, the Convention Establishing the World Intellectual Property Organization came into full force and officially established WIPO. In 1974, WIPO went on to become a specialized agency under the UN.
It offers a framework for cooperation between the member states in the field of intellectual property law, the protection of intellectual property rights and the registration of intellectual property.
Celebrate World Intellectual Property Day 2024 with themed quotes, images, messages, and more! Explore engaging content including posters, banners, and Instagram captions.
Best Quotes, Images, and Messages People recognize intellectual property the same way they recognize real estate. People understand what property is. But it’s a new kind of property, and so the understanding uses new control surfaces. It uses a new way of defining the property- Michael Nesmith
In the old days, you would have one lawyer to handle everything: speeding tickets, buying a house, contracts, litigation, real estate, copyrights, leasing, entertainment, intellectual property, forensic accounting, criminal offenses… the list goes on. Now, you have to have a separate lawyer for each one of those categories!- James Belushi
You cannot steal somebody’s intellectual property. Law and justice protect- Bikram Choudhury
Posters, Banners, Cliparts and Instagram Captions Globalisation, technological change, and the move to flexible labour markets has channelled more and more income to rentiers – those owning financial, physical, or so-called intellectual property – while real wages stagnate.- Guy Standing
“Innovation is the calling card of the future.” – Anna Eshoo
“Creativity is intelligence having fun.” – Albert Einstein
Why Emerald Gems Are Cool: Let’s See Published
19 hours ago on
April 25, 2024 Emeralds, with their captivating green hues, have held a special place in human history for millennia. From ancient civilizations to modern fashion, these gems have never lost their allure. But what makes emeralds so cool? Let’s delve into their unique characteristics and timeless appeal.
Firstly, let’s talk about color. The vibrant green of emeralds is unlike any other gemstone. It’s a color that symbolizes nature, growth, and harmony. The rich tones range from deep forest greens to lighter, more translucent shades, offering a variety that suits every taste and style. This diversity in color ensures that each emerald is as unique as the person wearing it.
Next, let’s consider their rarity. Emeralds are not as abundant as other gemstones, making them all the more precious. They are primarily found in a few select locations around the world, including Colombia, Zambia, and Brazil. The limited supply adds to their exclusivity and mystique, making them highly coveted by collectors and enthusiasts alike.
Another fascinating aspect of emeralds is their history and cultural significance. These gems have been prized for centuries by various civilizations, from the ancient Egyptians to the Incas. They have been revered for their supposed mystical properties, believed to bring wisdom, fertility, and good fortune to those who wear them. Throughout history, emeralds have adorned the crowns of royalty, the jewelry of nobility, and the amulets of spiritual leaders.
Moreover, Emerald Gemstone
Additionally, emeralds have a timeless appeal that transcends trends and fads. Unlike some gemstones that come in and out of fashion, emeralds have maintained their popularity throughout history. Their classic elegance makes them a staple in both traditional and contemporary jewelry designs. Whether paired with diamonds for a glamorous look or set in minimalist settings for a modern aesthetic, emeralds add a touch of sophistication to any ensemble.
Furthermore, emeralds have an undeniable allure that captures the imagination. There’s something mesmerizing about their intense green color and natural flaws, known as inclusions, which lend each stone its unique character. The way emeralds catch the light and seem to glow from within is truly enchanting, drawing the viewer into their depths.
In conclusion, emeralds are undeniably cool for a multitude of reasons. From their mesmerizing color and rarity to their rich history and enduring appeal, these gems continue to fascinate and captivate people around the world. Whether worn as a symbol of love, luck, or simply as a stunning accessory, emeralds are sure to remain cherished for generations to come. So, the next time you admire an emerald, take a moment to appreciate its beauty and the stories it holds within.
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(function(d, s, id) { var js, fjs = d.getElementsByTagName(s)[0]; if (d.getElementById(id)) return; js = d.createElement(s); js.id = id; js.src = "https://connect.facebook.net/en_US/all.js#xfbml=1&appId=178196885542208"; fjs.parentNode.insertBefore(js, fjs);}(document, 'script', 'facebook-jssdk'));
<p> $(document).ready(function(){
$('#commentbtn').on("click",function(){
(function(d, s, id) {
var js, fjs = d.getElementsByTagName(s)[0];
if (d.getElementById(id)) return;
js = d.createElement(s); js.id = id;
js.src = "https://connect.facebook.net/en_US/all.js#xfbml=1&appId=178196885542208";
fjs.parentNode.insertBefore(js, fjs);
}(document, 'script', 'facebook-jssdk'));
<p> $(".cmntbox").toggle();
});
});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
(adsbygoogle = window.adsbygoogle || []).push({});
if (window.innerWidth < 768) { jQuery(document).find('.ays-pb-modal_2').css({'top': '0', 'right': '0', 'bottom': '0', 'left': '0'}); } else { jQuery(document).find('.ays-pb-modal_2').css({'top': '0', 'right': '0', 'bottom': '0', 'left': '0'}); }
(function( $ ) {
'use strict';
$(document).ready(function(){
let pbViewsFlag_2 = true;
if ('image_type_img_theme' == 'notification') {
$(document).find('.ays-pb-modals').prependTo($(document.body));
} else {
$(document).find('.ays-pb-modals:not(.ays-pb-modals.ays-pb-notification-modal)').appendTo($(document.body));
}
let isMobile = false;
let closePopupOverlay = 1;
let isPageScrollDisabled = 0;
let checkAnimSpeed = 1;
let ays_pb_animation_close_speed = $(document).find('#ays_pb_animation_close_speed_2').val();
let ays_pb_effectIn_2 = $(document).find('#ays_pb_modal_animate_in_2').val();
let ays_pb_effectOut_2 = $(document).find('#ays_pb_modal_animate_out_2').val();
if (window.innerWidth < 768) {
isMobile = true;
closePopupOverlay = 1;
isPageScrollDisabled = 0;
checkAnimSpeed = 1;
ays_pb_animation_close_speed = $(document).find('#ays_pb_animation_close_speed_mobile_2').val();
ays_pb_effectIn_2 = $(document).find('#ays_pb_modal_animate_in_mobile_2').val();
ays_pb_effectOut_2 = $(document).find('#ays_pb_modal_animate_out_mobile_2').val();
}
let ays_pb_delayOpen_2 = parseInt($(document).find('.ays_pb_delay_2').val());
let ays_pb_scrollTop_2 = parseInt($(document).find('.ays_pb_scroll_2').val());
if (isMobile) {
if (0) {
ays_pb_scrollTop_2= parseInt($(document).find('.ays_pb_scroll_mobile_2').val());
}if (0) {
ays_pb_delayOpen_2 = parseInt($(document).find('.ays_pb_delay_mobile_2').val());
}
}
let time_pb_2 = $(document).find('.ays_pb_timer_2 span').data('seconds'),
ays_pb_animation_close_seconds = (ays_pb_animation_close_speed / 1000);
if( ays_pb_delayOpen_2 == 0 && ays_pb_scrollTop_2 == 0){
$(document).find('.av_pop_modals_2').css('display','block');
}if (window.innerWidth < 768) {
var mobileTimer = +$(document).find('.ays_pb_timer_2 span').attr('data-ays-mobile-seconds');
$(document).find('.ays_pb_timer_2 span').html(mobileTimer);
time_pb_2 = mobileTimer;
}ays_pb_animation_close_speed = parseFloat(ays_pb_animation_close_speed) - 50;$(document).find('.ays_music_sound').css({'display':'none'});
if(time_pb_2 !== undefined){
if(time_pb_2 !== 0){
$(document).find('#ays-pb-modal-checkbox_2').trigger('click');
if(ays_pb_scrollTop_2 == 0){
var ays_pb_flag = true;
$(document).find('.ays-pb-modal_2').css({
'animation-duration': ays_pb_animation_close_seconds + 's'
});
let timer_pb_2 = setInterval(function(){
let newTime_pb_2 = time_pb_2--;
$(document).find('.ays_pb_timer_2 span').text(newTime_pb_2);
if(newTime_pb_2 <= 0){
$(document).find('.ays-pb-modal-close_2').trigger('click');
$(document).find('.ays-pb-modal_2').attr('class', 'ays-pb-modal ays-pb-modal-image-type-img ays-pb-modal_2 ays-pb-border-mobile_2 '+ays_pb_effectOut_2);
if(ays_pb_effectOut_2 != 'none'){
setTimeout(function(){
$(document).find('.ays-pb-modal_2').css('display', 'none');
$(document).find('.ays-pb-modal_2').attr('data-ays-flag','true');
}, ays_pb_animation_close_speed);
}else{
$(document).find('.ays-pb-modal_2').css('display', 'none');
$(document).find('.ays-pb-modal_2').attr('data-ays-flag','true');
}
clearInterval(timer_pb_2);
}
$(document).find('.ays-pb-modal-close_2').one('click', function(){
if (pbViewsFlag_2) {
var pb_id = 2;$.ajax({
url: pbLocalizeObj.ajax,
method: 'POST',
dataType: 'text',
data: {
id: pb_id,
action: 'ays_increment_pb_views',
},
});pbViewsFlag_2 = false;
}
$(document).find('.av_pop_modals_2').css('pointer-events', 'none');
$(document).find('.ays-pb-modal_2').attr('class', 'ays-pb-modal ays-pb-modal-image-type-img ays-pb-modal_2 ays-pb-border-mobile_2 '+ays_pb_effectOut_2);
$(this).parents('.ays-pb-modals').find('iframe').each(function(){
var key = /https:\/\/www.youtube.com/;
var src = $(this).attr('src');
$(this).attr('src', $(this).attr('src'));
});
$(this).parents('.ays-pb-modals').find('video.wp-video-shortcode').each(function(){
if(typeof $(this).get(0) != 'undefined'){
if ( ! $(this).get(0).paused ) {
$(this).get(0).pause();
}
}
});
$(this).parents('.ays-pb-modals').find('audio.wp-audio-shortcode').each(function(){
if(typeof $(this).get(0) != 'undefined'){
if ( ! $(this).get(0).paused ) {
$(this).get(0).pause();
}
}
});
var close_sound_src = $(document).find('#ays_pb_close_sound_2').attr('src');
if(checkAnimSpeed && typeof close_sound_src !== 'undefined' && 'off' === 'on'){
if(checkAnimSpeed !== 0){
var playPromise = $(document).find('#ays_pb_close_sound_2').get(0).play();
if (playPromise !== undefined) {
playPromise.then(function() {
audio.pause();
}).catch(function(error) {
});
}
}
}
if(ays_pb_effectOut_2 != 'none'){
setTimeout(function(){
$(document).find('.ays-pb-modal_2').css('display', 'none');
$(document).find('.av_pop_modals_2').css('display', 'none');
$(document).find('.ays-pb-modal_2').attr('data-ays-flag','true');
if($('#ays_pb_close_sound_2').get(0) != undefined){
if(!$('#ays_pb_close_sound_2').get(0).paused){
$(document).find('.ays-pb-modal_2').css('display', 'none');
var audio = $('#ays_pb_close_sound_2').get(0);
audio.pause();
audio.currentTime = 0;
}
}
}, ays_pb_animation_close_speed);
}else{
$(document).find('.ays-pb-modal_2').css('display', 'none');
$(document).find('.av_pop_modals_2').css('display', 'none');
$(document).find('.ays-pb-modal_2').attr('data-ays-flag','true');
if($('#ays_pb_close_sound_2').get(0) != undefined){
if(!$('#ays_pb_close_sound_2').get(0).paused){
$(document).find('.ays-pb-modal_2').css('display', 'none');
var audio = $('#ays_pb_close_sound_2').get(0);
audio.pause();
audio.currentTime = 0;
}
}
}
$(document).find('#ays-pb-screen-shade_2').css({'opacity': '0', 'display': 'none'});
clearInterval(timer_pb_2);
});
$(document).on('keydown', function(event) {
if('1' && ays_pb_flag){
var escClosingPopups = $(document).find('.ays-pb-close-popup-with-esc:visible');
if (event.keyCode == 27) {
var topmostPopup = escClosingPopups.last();
topmostPopup.find('.ays-pb-modal-close_2').trigger('click');
}
} else {
ays_pb_flag = true;
}
ays_pb_flag = false;
});
},1000);
if(closePopupOverlay && 'On' == 'On'){
$(document).find('.av_pop_modals_2').on('click', function(e) {
var pb_parent = $(this);
var pb_div = $(this).find('.ays-pb-modal_2');
if (!pb_div.is(e.target) && pb_div.has(e.target).length === 0){
$(document).find('.ays-pb-modal-close_2').click();
}
});
}
}
} else {
$(document).find('.ays_pb_timer_2').css('display','none');
$(document).find('.ays-pb-modal_2').css({
'animation-duration': ays_pb_animation_close_seconds + 's'
});
$(document).find('.ays-pb-modal-close_2').one('click', function(){
if (pbViewsFlag_2) {
var pb_id = 2;$.ajax({
url: pbLocalizeObj.ajax,
method: 'POST',
dataType: 'text',
data: {
id: pb_id,
action: 'ays_increment_pb_views',
},
});pbViewsFlag_2 = false;
}
$(document).find('.av_pop_modals_2').css('pointer-events', 'none');
$(document).find('.ays-pb-modal_2').attr('class', 'ays-pb-modal ays-pb-modal-image-type-img ays-pb-modal_2 ays-pb-border-mobile_2 '+ays_pb_effectOut_2);
$(this).parents('.ays-pb-modals').find('iframe').each(function(){
var key = /https:\/\/www.youtube.com/;
var src = $(this).attr('src');
$(this).attr('src', $(this).attr('src'));
});
$(this).parents('.ays-pb-modals').find('video.wp-video-shortcode').each(function(){
if(typeof $(this).get(0) != 'undefined'){
if ( ! $(this).get(0).paused ) {
$(this).get(0).pause();
}
}
});
$(this).parents('.ays-pb-modals').find('audio.wp-audio-shortcode').each(function(){
if(typeof $(this).get(0) != 'undefined'){
if ( ! $(this).get(0).paused ) {
$(this).get(0).pause();
}
}
});
if(ays_pb_effectOut_2 != 'none'){
setTimeout(function(){
$(document).find('.ays-pb-modal_2').css('display', 'none');
$(document).find('.av_pop_modals_2').css('display', 'none');
$(document).find('.ays-pb-modal_2').attr('data-ays-flag','true');
if($('#ays_pb_close_sound_2').get(0) != undefined){
if(!$('#ays_pb_close_sound_2').get(0).paused){
$(document).find('.ays-pb-modal_2').css('display', 'none');
var audio = $('#ays_pb_close_sound_2').get(0);
audio.pause();
audio.currentTime = 0;
}
}
}, ays_pb_animation_close_speed);
}else{
$(document).find('.ays-pb-modal_2').css('display', 'none');
$(document).find('.av_pop_modals_2').css('display', 'none');
$(document).find('.ays-pb-modal_2').attr('data-ays-flag','true');
if($('#ays_pb_close_sound_2').get(0) != undefined){
if(!$('#ays_pb_close_sound_2').get(0).paused){
$(document).find('.ays-pb-modal_2').css('display', 'none');
var audio = $('#ays_pb_close_sound_2').get(0);
audio.pause();
audio.currentTime = 0;
}
}
}
$(document).find('#ays-pb-screen-shade_2').css({'opacity': '0', 'display': 'none'});
});
}
}
let count = 0;
if( ays_pb_scrollTop_2 !== 0 ){
$(window).scroll(function() {
if(($(this).scrollTop() >= ays_pb_scrollTop_2) && (count === 0)) {
count++;
if( ays_pb_delayOpen_2 !== 0 ){
$(document).find('.ays-pb-modal_2').css('animation-delay', ays_pb_delayOpen_2/1000);
setTimeout(function(){
$(document).find('.av_pop_modals_2').css('display','block');
$(document).find('.ays-pb-modal_2').addClass(ays_pb_effectIn_2);
$(document).find('.ays-pb-modal_2').css('display', 'block');
if (window.innerWidth < 768 && $(document).find('#ays-pb-screen-shade_2').attr('data-mobile-overlay') == 'true') {
$(document).find('#ays-pb-screen-shade_2').css({'opacity': '0.5'});
}
else{
$(document).find('#ays-pb-screen-shade_2').css({'opacity': '0.500000'});
}$(document).find('.ays-pb-modal-check_2').prop('checked', true);
}, ays_pb_delayOpen_2);
}else{
$(document).find('.av_pop_modals_2').css('display','block');
$(document).find('.ays-pb-modal_2').addClass(ays_pb_effectIn_2);
$(document).find('.ays-pb-modal_2').css('display', 'block');
if (window.innerWidth < 768 && $(document).find('#ays-pb-screen-shade_2').attr('data-mobile-overlay') == 'true') {
$(document).find('#ays-pb-screen-shade_2').css({'opacity': '0.5'});
}
else{
$(document).find('#ays-pb-screen-shade_2').css({'opacity': '0.500000'});
}$(document).find('.ays-pb-modal-check_2').prop('checked', true);
}
if('false' == 'true' && window.innerWidth < 768){
if(0 != 0 && 'off' != 'on'){
let close_button_delay_for_mobile = 0;
if (ays_pb_delayOpen_2 != 0) {
close_button_delay_for_mobile += Math.floor(ays_pb_delayOpen_2);
}
$(document).find('.ays-pb-modal-close_2').css({'display': 'none'});
setTimeout(function(){
$(document).find('.ays-pb-modal-close_2').css({'display': 'block'});
}, close_button_delay_for_mobile );
}
}
else {
if(0 != 0 && 'off' != 'on'){
let close_button_delay = 0;
if (ays_pb_delayOpen_2 != 0) {
close_button_delay += Math.floor(ays_pb_delayOpen_2);
}
$(document).find('.ays-pb-modal-close_2').css({'display': 'none'});
setTimeout(function(){
$(document).find('.ays-pb-modal-close_2').css({'display': 'block'});
}, close_button_delay );
}
}
if(5 != 0){
$(document).find('.ays-pb-modal_2').css({
'animation-duration': ays_pb_animation_close_seconds + 's'
});
let timer_pb_2 = setInterval(function(){
let newTime_pb_2 = time_pb_2--;
$(document).find('.ays_pb_timer_2 span').text(newTime_pb_2);
if(newTime_pb_2 <= 0){
$(document).find('.ays-pb-modal-close_2').trigger('click');
$(document).find('.ays-pb-modal_2').attr('class', 'ays-pb-modal ays-pb-modal-image-type-img ays-pb-modal_2 '+ays_pb_effectOut_2);
if(ays_pb_effectOut_2 != 'none'){
setTimeout(function(){
$(document).find('.ays-pb-modal_2').css('display', 'none');
$(document).find('.ays-pb-modal_2').attr('data-ays-flag','true');
}, ays_pb_animation_close_speed);
}else{
$(document).find('.ays-pb-modal_2').css('display', 'none');
$(document).find('.ays-pb-modal_2').attr('data-ays-flag','true');
}
clearInterval(timer_pb_2);
}
$(document).find('.ays-pb-modal-close_2').one('click', function(){
if (pbViewsFlag_2) {
var pb_id = 2;$.ajax({
url: pbLocalizeObj.ajax,
method: 'POST',
dataType: 'text',
data: {
id: pb_id,
action: 'ays_increment_pb_views',
},
});pbViewsFlag_2 = false;
}
$(document).find('.av_pop_modals_2').css('pointer-events', 'none');
$(document).find('.ays-pb-modal_2').attr('class', 'ays-pb-modal ays-pb-modal-image-type-img ays-pb-modal_2 ays-pb-border-mobile_2 '+ays_pb_effectOut_2);
$(this).parents('.ays-pb-modals').find('iframe').each(function(){
var key = /https:\/\/www.youtube.com/;
var src = $(this).attr('src');
$(this).attr('src', $(this).attr('src'));
});
$(this).parents('.ays-pb-modals').find('video.wp-video-shortcode').each(function(){
if(typeof $(this).get(0) != 'undefined'){
if ( ! $(this).get(0).paused ) {
$(this).get(0).pause();
}
}
});
$(this).parents('.ays-pb-modals').find('audio.wp-audio-shortcode').each(function(){
if(typeof $(this).get(0) != 'undefined'){
if ( ! $(this).get(0).paused ) {
$(this).get(0).pause();
}
}
});
if(ays_pb_effectOut_2 != 'none'){
setTimeout(function(){
$(document).find('.ays-pb-modal_2').css('display', 'none');
$(document).find('.av_pop_modals_2').css('display', 'none');
$(document).find('.ays-pb-modal_2').attr('data-ays-flag','true');
if($('#ays_pb_close_sound_2').get(0) != undefined){
if(!$('#ays_pb_close_sound_2').get(0).paused){
$(document).find('.ays-pb-modal_2').css('display', 'none');
var audio = $('#ays_pb_close_sound_2').get(0);
audio.pause();
audio.currentTime = 0;
}
}
}, ays_pb_animation_close_speed);
}else{
$(document).find('.ays-pb-modal_2').css('display', 'none');
$(document).find('.av_pop_modals_2').css('display', 'none');
$(document).find('.ays-pb-modal_2').attr('data-ays-flag','true');
if($('#ays_pb_close_sound_2').get(0) != undefined){
if(!$('#ays_pb_close_sound_2').get(0).paused){
$(document).find('.ays-pb-modal_2').css('display', 'none');
var audio = $('#ays_pb_close_sound_2').get(0);
audio.pause();
audio.currentTime = 0;
}
}
}
$(document).find('#ays-pb-screen-shade_2').css({'opacity': '0', 'display': 'none'});
clearInterval(timer_pb_2);
});
var ays_pb_flag = true;
$(document).on('keydown', function(event) {
if('1' && ays_pb_flag){
var escClosingPopups = $(document).find('.ays-pb-close-popup-with-esc:visible');
if (event.keyCode == 27) {
var topmostPopup = escClosingPopups.last();
topmostPopup.find('.ays-pb-modal-close_2').trigger('click');
ays_pb_flag = false;
}
} else {
ays_pb_flag = true;
}
});
},1000);
}
}
});
}else{
if( ays_pb_delayOpen_2 !== 0 ){
$(document).find('.ays-pb-modal_2').css('animation-delay', ays_pb_delayOpen_2/1000);
setTimeout(function(){
$(document).find('.av_pop_modals_2').css('display','block');
$(document).find('.ays-pb-modal_2').addClass(ays_pb_effectIn_2);
$(document).find('.ays-pb-modal_2').css('display', 'block');
if (window.innerWidth < 768 && $(document).find('#ays-pb-screen-shade_2').attr('data-mobile-overlay') == 'true') {
$(document).find('#ays-pb-screen-shade_2').css({'opacity': '0.5'});
}
else{
$(document).find('#ays-pb-screen-shade_2').css({'opacity': '0.500000'});
}
$(document).find('.ays-pb-modal-check_2').attr('checked', 'checked');if(isPageScrollDisabled){
$(document).find('body').addClass('pb_disable_scroll_2');
$(document).find('html').removeClass('pb_enable_scroll');
}}, ays_pb_delayOpen_2);
} else {
if($(document).find('.ays_pb_abt_2').val() != 'clickSelector'){
$(document).find('.av_pop_modals_2').css('display','block');
$(document).find('.ays-pb-modal_2').addClass(ays_pb_effectIn_2);
$(document).find('.ays-pb-modal_2').css('display', 'block');
if (window.innerWidth < 768 && $(document).find('#ays-pb-screen-shade_2').attr('data-mobile-overlay') == 'true') {
$(document).find('#ays-pb-screen-shade_2').css({'opacity': '0.5'});
}
else{
$(document).find('#ays-pb-screen-shade_2').css({'opacity': '0.500000'});
}
$(document).find('.ays-pb-modal-check_2').attr('checked', 'checked');if(isPageScrollDisabled){
$(document).find('body').addClass('pb_disable_scroll_2');
$(document).find('html').addClass('pb_disable_scroll_2');
}
}
}
}
if ('On' != 'On'){
$(document).find('#ays-pb-screen-shade_2').css({'opacity': '0', 'display': 'none !important', 'pointer-events': 'none', 'background': 'none'});
$(document).find('.ays-pb-modal_2').css('pointer-events', 'auto');
$(document).find('.av_pop_modals_2').css('pointer-events','none');
};
if($(document).find('.ays-pb-modals video').hasClass('wp-video-shortcode')){
var videoWidth = $(document).find('.ays-pb-modals video.wp-video-shortcode').attr('width');
var videoHeight = $(document).find('.ays-pb-modals video.wp-video-shortcode').attr('height');
setTimeout(function(){
$(document).find('.ays-pb-modals .wp-video').removeAttr('style');
$(document).find('.ays-pb-modals .mejs-container').removeAttr('style');
$(document).find('.ays-pb-modals video.wp-video-shortcode').removeAttr('style');$(document).find('.ays-pb-modals .wp-video').css({'width': '100%'});
$(document).find('.ays-pb-modals .mejs-container').css({'width': '100%','height': videoHeight + 'px'});
$(document).find('.ays-pb-modals video.wp-video-shortcode').css({'width': '100%','height': videoHeight + 'px'});
},1000);
}
if($(document).find('.ays-pb-modals iframe').attr('style') != ''){
setTimeout(function(){
$(document).find('.ays-pb-modals iframe').removeAttr('style');
},500);
}
// if(5 == 0){
if(closePopupOverlay && 'On' == 'On'){
$(document).find('.av_pop_modals_2').on('click', function(e) {
var pb_parent = $(this);
var pb_div = $(this).find('.ays-pb-modal_2');
if (!pb_div.is(e.target) && pb_div.has(e.target).length === 0){
$(document).find('.ays-pb-modal-close_2').click();
}
});
}
var ays_pb_flag = true;
$(document).on('keydown', function(event) {
if('1' && ays_pb_flag){
var escClosingPopups = $(document).find('.ays-pb-close-popup-with-esc:visible');
if (event.keyCode == 27) {
var topmostPopup = escClosingPopups.last();
topmostPopup.find('.ays-pb-modal-close_2').trigger('click');
ays_pb_flag = false;
}
} else {
ays_pb_flag = true;
}
});
// }
if('off' == 'on') {
var video = $(document).find('video.wp-video-shortcode');
for (let i = 0; i < video.length; i++) {
video[i].addEventListener('ended', function() {
if ($(this).next().val() === 'on') {
$(this).parents('.ays_video_window').find('.close-image-btn').trigger('click');
}
});
}
}
jQuery(document).on('click', '.ays-pb-modal-close_2', function() {
$(document).find('body').removeClass('pb_disable_scroll_2');
$(document).find('html').removeClass('pb_disable_scroll_2');
});});
})( jQuery );
if(typeof aysPopupOptions === "undefined"){
var aysPopupOptions = [];
}
aysPopupOptions["2"] = "eyJwb3B1cGJveCI6eyJpZCI6IjIiLCJ0aXRsZSI6IkFkIiwicG9wdXBfbmFtZSI6IiIsImRlc2NyaXB0aW9uIjoiRGVtbyBEZXNjcmlwdGlvbiIsImNhdGVnb3J5X2lkIjoiMSIsImF1dG9jbG9zZSI6IjUiLCJjb29raWUiOiIxNDQwIiwid2lkdGgiOjQwMCwiaGVpZ2h0Ijo1MDAsImJnY29sb3IiOiIjZmZmZmZmIiwidGV4dGNvbG9yIjoiIzAwMDAwMCIsImJvcmRlcnNpemUiOiIxIiwiYm9yZGVyY29sb3IiOiIjZmZmZmZmIiwiYm9yZGVyX3JhZGl1cyI6IjQiLCJzaG9ydGNvZGUiOiIiLCJ1c2Vyc19yb2xlIjoiW10iLCJjdXN0b21fY2xhc3MiOiIiLCJjdXN0b21fY3NzIjoiIiwiY3VzdG9tX2h0bWwiOiJIZXJlIGNhbiBiZSB5b3VyIGN1c3RvbSBIVE1MIG9yIFNob3J0Y29kZSIsIm9ub2Zmc3dpdGNoIjoiT24iLCJzaG93X29ubHlfZm9yX2F1dGhvciI6Im9mZiIsInNob3dfYWxsIjoiYWxsIiwiZGVsYXkiOiIzIiwic2Nyb2xsX3RvcCI6IjAiLCJhbmltYXRlX2luIjoiZmFkZUluIiwiYW5pbWF0ZV9vdXQiOiJmYWRlT3V0IiwiYWN0aW9uX2J1dHRvbiI6IiIsInZpZXdfcGxhY2UiOiIiLCJhY3Rpb25fYnV0dG9uX3R5cGUiOiJwYWdlTG9hZGVkIiwibW9kYWxfY29udGVudCI6ImltYWdlX3R5cGUiLCJ2aWV3X3R5cGUiOiJpbWFnZV90eXBlX2ltZ190aGVtZSIsIm9ub2Zmb3ZlcmxheSI6Ik9uIiwib3ZlcmxheV9vcGFjaXR5IjoiMC41MDAwMDAiLCJzaG93X3BvcHVwX3RpdGxlIjoiT2ZmIiwic2hvd19wb3B1cF9kZXNjIjoiT2ZmIiwiY2xvc2VfYnV0dG9uIjoib2ZmIiwiaGVhZGVyX2JnY29sb3IiOiIjZmZmZmZmIiwiYmdfaW1hZ2UiOiIiLCJsb2dfdXNlciI6Ik9uIiwiZ3Vlc3QiOiJPbiIsImFjdGl2ZV9kYXRlX2NoZWNrIjoib2ZmIiwiYWN0aXZlSW50ZXJ2YWwiOiIyMDI0LTA0LTE5IDE2OjA0OjE3IiwiZGVhY3RpdmVJbnRlcnZhbCI6IjIwMjQtMDQtMTkgMTY6MDQ6MTciLCJwYl9wb3NpdGlvbiI6ImNlbnRlci1jZW50ZXIiLCJwYl9tYXJnaW4iOiIwIiwidmlld3MiOiIzMjM4MCIsImNvbnZlcnNpb25zIjoiMCIsIm9wdGlvbnMiOiJ7XCJlbmFibGVfYmFja2dyb3VuZF9ncmFkaWVudFwiOlwib2ZmXCIsXCJiYWNrZ3JvdW5kX2dyYWRpZW50X2NvbG9yXzFcIjpcIiMwMDBcIixcImJhY2tncm91bmRfZ3JhZGllbnRfY29sb3JfMlwiOlwiI2ZmZlwiLFwicGJfZ3JhZGllbnRfZGlyZWN0aW9uXCI6XCJ2ZXJ0aWNhbFwiLFwiZW5hYmxlX2JhY2tncm91bmRfZ3JhZGllbnRfbW9iaWxlXCI6XCJvZmZcIixcImJhY2tncm91bmRfZ3JhZGllbnRfY29sb3JfMV9tb2JpbGVcIjpcIiMwMDBcIixcImJhY2tncm91bmRfZ3JhZGllbnRfY29sb3JfMl9tb2JpbGVcIjpcIiNmZmZcIixcInBiX2dyYWRpZW50X2RpcmVjdGlvbl9tb2JpbGVcIjpcInZlcnRpY2FsXCIsXCJleGNlcHRfcG9zdF90eXBlc1wiOltdLFwiZXhjZXB0X3Bvc3RzXCI6W10sXCJhbGxfcG9zdHNcIjpcIlwiLFwiY2xvc2VfYnV0dG9uX2RlbGF5XCI6MCxcImNsb3NlX2J1dHRvbl9kZWxheV9mb3JfbW9iaWxlXCI6MCxcImVuYWJsZV9jbG9zZV9idXR0b25fZGVsYXlfZm9yX21vYmlsZVwiOlwib2ZmXCIsXCJlbmFibGVfcGJfc291bmRcIjpcIm9mZlwiLFwib3ZlcmxheV9jb2xvclwiOlwiIzAwMFwiLFwiZW5hYmxlX292ZXJsYXlfY29sb3JfbW9iaWxlXCI6XCJvZmZcIixcIm92ZXJsYXlfY29sb3JfbW9iaWxlXCI6XCIjMDAwXCIsXCJhbmltYXRpb25fc3BlZWRcIjoxLFwiZW5hYmxlX2FuaW1hdGlvbl9zcGVlZF9tb2JpbGVcIjpcIm9mZlwiLFwiYW5pbWF0aW9uX3NwZWVkX21vYmlsZVwiOjEsXCJjbG9zZV9hbmltYXRpb25fc3BlZWRcIjoxLFwiZW5hYmxlX2Nsb3NlX2FuaW1hdGlvbl9zcGVlZF9tb2JpbGVcIjpcIm9mZlwiLFwiY2xvc2VfYW5pbWF0aW9uX3NwZWVkX21vYmlsZVwiOjEsXCJwYl9tb2JpbGVcIjpcIm9mZlwiLFwiY2xvc2VfYnV0dG9uX3RleHRcIjpcIlxcdTI3MTVcIixcImVuYWJsZV9jbG9zZV9idXR0b25fdGV4dF9tb2JpbGVcIjpcIm9uXCIsXCJjbG9zZV9idXR0b25fdGV4dF9tb2JpbGVcIjpcIlxcdTI3MTVcIixcImNsb3NlX2J1dHRvbl9ob3Zlcl90ZXh0XCI6XCJcIixcIm1vYmlsZV93aWR0aFwiOlwiXCIsXCJtb2JpbGVfbWF4X3dpZHRoXCI6XCJcIixcIm1vYmlsZV9oZWlnaHRcIjpcIlwiLFwiY2xvc2VfYnV0dG9uX3Bvc2l0aW9uXCI6XCJyaWdodC10b3BcIixcImVuYWJsZV9jbG9zZV9idXR0b25fcG9zaXRpb25fbW9iaWxlXCI6XCJvZmZcIixcImNsb3NlX2J1dHRvbl9wb3NpdGlvbl9tb2JpbGVcIjpcInJpZ2h0LXRvcFwiLFwic2hvd19vbmx5X29uY2VcIjpcIm9mZlwiLFwic2hvd19vbl9ob21lX3BhZ2VcIjpcIm9mZlwiLFwiY2xvc2VfcG9wdXBfZXNjXCI6XCJvblwiLFwicG9wdXBfd2lkdGhfYnlfcGVyY2VudGFnZV9weFwiOlwicGl4ZWxzXCIsXCJwb3B1cF9jb250ZW50X3BhZGRpbmdcIjowLFwicG9wdXBfcGFkZGluZ19ieV9wZXJjZW50YWdlX3B4XCI6XCJwaXhlbHNcIixcInBiX2ZvbnRfZmFtaWx5XCI6XCJpbmhlcml0XCIsXCJjbG9zZV9wb3B1cF9vdmVybGF5XCI6XCJvblwiLFwiY2xvc2VfcG9wdXBfb3ZlcmxheV9tb2JpbGVcIjpcIm9uXCIsXCJlbmFibGVfcGJfZnVsbHNjcmVlblwiOlwib2ZmXCIsXCJlbmFibGVfaGlkZV90aW1lclwiOlwib2ZmXCIsXCJlbmFibGVfaGlkZV90aW1lcl9tb2JpbGVcIjpcIm9mZlwiLFwiZW5hYmxlX2F1dG9jbG9zZV9vbl9jb21wbGV0aW9uXCI6XCJvZmZcIixcImVuYWJsZV9zb2NpYWxfbGlua3NcIjpcIm9mZlwiLFwic29jaWFsX2xpbmtzXCI6e1wibGlua2VkaW5fbGlua1wiOlwiXCIsXCJmYWNlYm9va19saW5rXCI6XCJcIixcInR3aXR0ZXJfbGlua1wiOlwiXCIsXCJ2a29udGFrdGVfbGlua1wiOlwiXCIsXCJ5b3V0dWJlX2xpbmtcIjpcIlwiLFwiaW5zdGFncmFtX2xpbmtcIjpcIlwiLFwiYmVoYW5jZV9saW5rXCI6XCJcIn0sXCJzb2NpYWxfYnV0dG9uc19oZWFkaW5nXCI6XCJcIixcImNsb3NlX2J1dHRvbl9zaXplXCI6MSxcImNsb3NlX2J1dHRvbl9pbWFnZVwiOlwiXCIsXCJib3JkZXJfc3R5bGVcIjpcInNvbGlkXCIsXCJlbmFibGVfYm9yZGVyX3N0eWxlX21vYmlsZVwiOlwib2ZmXCIsXCJib3JkZXJfc3R5bGVfbW9iaWxlXCI6XCJzb2xpZFwiLFwiYXlzX3BiX2hvdmVyX3Nob3dfY2xvc2VfYnRuXCI6XCJvZmZcIixcImRpc2FibGVfc2Nyb2xsXCI6XCJvZmZcIixcImRpc2FibGVfc2Nyb2xsX21vYmlsZVwiOlwib2ZmXCIsXCJlbmFibGVfb3Blbl9kZWxheV9tb2JpbGVcIjpcIm9mZlwiLFwib3Blbl9kZWxheV9tb2JpbGVcIjpcIjBcIixcImVuYWJsZV9zY3JvbGxfdG9wX21vYmlsZVwiOlwib2ZmXCIsXCJzY3JvbGxfdG9wX21vYmlsZVwiOlwiMFwiLFwiZW5hYmxlX3BiX3Bvc2l0aW9uX21vYmlsZVwiOlwib2ZmXCIsXCJwYl9wb3NpdGlvbl9tb2JpbGVcIjpcImNlbnRlci1jZW50ZXJcIixcInBiX2JnX2ltYWdlX3Bvc2l0aW9uXCI6XCJjZW50ZXItY2VudGVyXCIsXCJlbmFibGVfcGJfYmdfaW1hZ2VfcG9zaXRpb25fbW9iaWxlXCI6XCJvZmZcIixcInBiX2JnX2ltYWdlX3Bvc2l0aW9uX21vYmlsZVwiOlwiY2VudGVyLWNlbnRlclwiLFwicGJfYmdfaW1hZ2Vfc2l6aW5nXCI6XCJjb3ZlclwiLFwiZW5hYmxlX3BiX2JnX2ltYWdlX3NpemluZ19tb2JpbGVcIjpcIm9mZlwiLFwicGJfYmdfaW1hZ2Vfc2l6aW5nX21vYmlsZVwiOlwiY292ZXJcIixcInZpZGVvX3RoZW1lX3VybFwiOlwiXCIsXCJpbWFnZV90eXBlX2ltZ19zcmNcIjpcImh0dHBzOlxcXC9cXFwvd3d3LnVuaXF1ZW5ld3NvbmxpbmUuY29tXFxcL3dwLWNvbnRlbnRcXFwvdXBsb2Fkc1xcXC8yMDI0XFxcLzA0XFxcL2Jhbm5lci1zYXRzc3BvcnRzLmpwZ1wiLFwiaW1hZ2VfdHlwZV9pbWdfcmVkaXJlY3RfdXJsXCI6XCJodHRwczpcXFwvXFxcL3d3dy5zYXRzcG9ydC5jb21cXFwveFxcXC8jXFxcLzJcXFwvaG9tZVxcXC9leGNoYW5nZVxcXC9zcG9ydFxcXC9hbGw/c2lnbnVwPXRydWUmdXRtX3NvdXJjZT11bmlxdWVuZXdzb25saW5lJnV0bV9tZWRpdW09dW5pcXVlbmV3c29ubGluZVwiLFwiZmFjZWJvb2tfcGFnZV91cmxcIjpcImh0dHBzOlxcXC9cXFwvd3d3LmZhY2Vib29rLmNvbVxcXC93b3JkcHJlc3NcIixcImhpZGVfZmJfcGFnZV9jb3Zlcl9waG90b1wiOlwib2ZmXCIsXCJub3RpZmljYXRpb25fdHlwZV9jb21wb25lbnRzXCI6W10sXCJub3RpZmljYXRpb25fdHlwZV9jb21wb25lbnRzX29yZGVyXCI6e1wibWFpbl9jb250ZW50XCI6XCJtYWluX2NvbnRlbnRcIixcImJ1dHRvbl8xXCI6XCJidXR0b25fMVwifSxcIm5vdGlmaWNhdGlvbl9tYWluX2NvbnRlbnRcIjpcIldyaXRlIHRoZSBjdXN0b20gbm90aWZpY2F0aW9uIGJhbm5lciB0ZXh0IGhlcmUuXCIsXCJub3RpZmljYXRpb25fYnV0dG9uXzFfdGV4dFwiOlwiQ2xpY2shXCIsXCJub3RpZmljYXRpb25fYnV0dG9uXzFfcmVkaXJlY3RfdXJsXCI6XCJcIixcInBiX21heF9oZWlnaHRcIjpcIlwiLFwicG9wdXBfbWF4X2hlaWdodF9ieV9wZXJjZW50YWdlX3B4XCI6XCJwaXhlbHNcIixcInBiX21heF9oZWlnaHRfbW9iaWxlXCI6XCJcIixcInBvcHVwX21heF9oZWlnaHRfYnlfcGVyY2VudGFnZV9weF9tb2JpbGVcIjpcInBpeGVsc1wiLFwicGJfbWluX2hlaWdodFwiOlwiXCIsXCJwYl9mb250X3NpemVcIjoxMyxcInBiX2ZvbnRfc2l6ZV9mb3JfbW9iaWxlXCI6MTMsXCJwYl90aXRsZV90ZXh0X3NoYWRvd1wiOlwicmdiYSgyNTUsMjU1LDI1NSwwKVwiLFwiZW5hYmxlX3BiX3RpdGxlX3RleHRfc2hhZG93XCI6XCJvZmZcIixcInBiX3RpdGxlX3RleHRfc2hhZG93X3hfb2Zmc2V0XCI6MixcInBiX3RpdGxlX3RleHRfc2hhZG93X3lfb2Zmc2V0XCI6MixcInBiX3RpdGxlX3RleHRfc2hhZG93X3pfb2Zmc2V0XCI6MCxcInBiX3RpdGxlX3RleHRfc2hhZG93X21vYmlsZVwiOlwicmdiYSgyNTUsMjU1LDI1NSwwKVwiLFwiZW5hYmxlX3BiX3RpdGxlX3RleHRfc2hhZG93X21vYmlsZVwiOlwib2ZmXCIsXCJwYl90aXRsZV90ZXh0X3NoYWRvd194X29mZnNldF9tb2JpbGVcIjoyLFwicGJfdGl0bGVfdGV4dF9zaGFkb3dfeV9vZmZzZXRfbW9iaWxlXCI6MixcInBiX3RpdGxlX3RleHRfc2hhZG93X3pfb2Zmc2V0X21vYmlsZVwiOjAsXCJjcmVhdGVfZGF0ZVwiOlwiMjAyNC0wNC0xNyAyMDoyOTo1NlwiLFwiY3JlYXRlX2F1dGhvclwiOjMsXCJhdXRob3JcIjpcIntcXFwiaWRcXFwiOlxcXCIzXFxcIixcXFwibmFtZVxcXCI6XFxcIkRyLiBZb2dlbmRyYSBEZXN3YXJcXFwifVwiLFwiZW5hYmxlX2Rpc21pc3NcIjpcIm9mZlwiLFwiZW5hYmxlX2Rpc21pc3NfdGV4dFwiOlwiRGlzbWlzcyBhZFwiLFwiZW5hYmxlX2Rpc21pc3NfbW9iaWxlXCI6XCJvZmZcIixcImVuYWJsZV9kaXNtaXNzX3RleHRfbW9iaWxlXCI6XCJEaXNtaXNzIGFkXCIsXCJlbmFibGVfYm94X3NoYWRvd1wiOlwib2ZmXCIsXCJlbmFibGVfYm94X3NoYWRvd19tb2JpbGVcIjpcIm9mZlwiLFwiYm94X3NoYWRvd19jb2xvclwiOlwiIzAwMFwiLFwiYm94X3NoYWRvd19jb2xvcl9tb2JpbGVcIjpcIiMwMDBcIixcInBiX2JveF9zaGFkb3dfeF9vZmZzZXRcIjowLFwicGJfYm94X3NoYWRvd194X29mZnNldF9tb2JpbGVcIjowLFwicGJfYm94X3NoYWRvd195X29mZnNldFwiOjAsXCJwYl9ib3hfc2hhZG93X3lfb2Zmc2V0X21vYmlsZVwiOjAsXCJwYl9ib3hfc2hhZG93X3pfb2Zmc2V0XCI6MTUsXCJwYl9ib3hfc2hhZG93X3pfb2Zmc2V0X21vYmlsZVwiOjE1LFwiZGlzYWJsZV9zY3JvbGxfb25fcG9wdXBcIjpcIm9mZlwiLFwiZGlzYWJsZV9zY3JvbGxfb25fcG9wdXBfbW9iaWxlXCI6XCJvZmZcIixcInNob3dfc2Nyb2xsYmFyXCI6XCJvZmZcIixcImhpZGVfb25fcGNcIjpcIm9mZlwiLFwiaGlkZV9vbl90YWJsZXRzXCI6XCJvZmZcIixcInBiX2JnX2ltYWdlX2RpcmVjdGlvbl9vbl9tb2JpbGVcIjpcIm9uXCIsXCJjbG9zZV9idXR0b25fY29sb3JcIjpcIiMwMDAwMDBcIixcImNsb3NlX2J1dHRvbl9ob3Zlcl9jb2xvclwiOlwiIzAwMDAwMFwiLFwiYmx1cmVkX292ZXJsYXlcIjpcIm9mZlwiLFwiYmx1cmVkX292ZXJsYXlfbW9iaWxlXCI6XCJvZmZcIixcInBiX2F1dG9jbG9zZV9tb2JpbGVcIjpcIjBcIixcImVuYWJsZV9hdXRvY2xvc2VfZGVsYXlfdGV4dF9tb2JpbGVcIjpcIm9mZlwiLFwiZW5hYmxlX292ZXJsYXlfdGV4dF9tb2JpbGVcIjpcIm9mZlwiLFwib3ZlcmxheV9tb2JpbGVfb3BhY2l0eVwiOlwiMC41XCIsXCJzaG93X3BvcHVwX3RpdGxlX21vYmlsZVwiOlwiT2ZmXCIsXCJzaG93X3BvcHVwX2Rlc2NfbW9iaWxlXCI6XCJPZmZcIixcImVuYWJsZV9hbmltYXRlX2luX21vYmlsZVwiOlwib2ZmXCIsXCJhbmltYXRlX2luX21vYmlsZVwiOlwiZmFkZUluXCIsXCJlbmFibGVfYW5pbWF0ZV9vdXRfbW9iaWxlXCI6XCJvZmZcIixcImFuaW1hdGVfb3V0X21vYmlsZVwiOlwiZmFkZU91dFwiLFwiZW5hYmxlX2Rpc3BsYXlfY29udGVudF9tb2JpbGVcIjpcIm9mZlwiLFwiZW5hYmxlX2JnY29sb3JfbW9iaWxlXCI6XCJvZmZcIixcImJnY29sb3JfbW9iaWxlXCI6XCIjZmZmZmZmXCIsXCJlbmFibGVfYmdfaW1hZ2VfbW9iaWxlXCI6XCJvZmZcIixcImJnX2ltYWdlX21vYmlsZVwiOlwiXCIsXCJlbmFibGVfYm9yZGVyY29sb3JfbW9iaWxlXCI6XCJvZmZcIixcImJvcmRlcmNvbG9yX21vYmlsZVwiOlwiI2ZmZmZmZlwiLFwiZW5hYmxlX2JvcmRlcnNpemVfbW9iaWxlXCI6XCJvZmZcIixcImJvcmRlcnNpemVfbW9iaWxlXCI6XCIxXCIsXCJlbmFibGVfYm9yZGVyX3JhZGl1c19tb2JpbGVcIjpcIm9mZlwiLFwiYm9yZGVyX3JhZGl1c19tb2JpbGVcIjpcIjRcIn0iLCJzaG93X3BvcHVwX3RpdGxlX21vYmlsZSI6Ik9mZiIsInNob3dfcG9wdXBfZGVzY19tb2JpbGUiOiJPZmYifX0=";
!function(){const e=document.createElement("script");e.async=!0,e.setAttribute("crossorigin","anonymous"),e.src="//pagead2.googlesyndication.com/pagead/js/adsbygoogle.js?client=ca-pub-9548258067481809";let t=0;document.addEventListener("mousemove",function(){1==++t&&document.getElementsByTagName("HEAD").item(0).appendChild(e)}),window.onscroll=function(n){1==++t&&document.getElementsByTagName("HEAD").item(0).appendChild(e)},setTimeout(function(){0===t&&(t++,document.getElementsByTagName("HEAD").item(0).appendChild(e))},5e3)}();
https://www.uniquenewsonline.com/wp-content/plugins/theia-sticky-sidebar/js/ResizeSensor.js
https://www.uniquenewsonline.com/wp-content/plugins/theia-sticky-sidebar/js/theia-sticky-sidebar.js
https://www.uniquenewsonline.com/wp-content/plugins/theia-sticky-sidebar/js/main.js
https://www.uniquenewsonline.com/wp-content/themes/zox-news/js/mvpcustom.js
jQuery(document).ready(function($) {
var leaderHeight = $("#mvp-leader-wrap").outerHeight();
var navHeight = $("#mvp-main-head-wrap").outerHeight();
var headerHeight = navHeight + leaderHeight;
var previousScroll = 0;
$(window).scroll(function(event){
var scroll = $(this).scrollTop();
if ( typeof leaderHeight !== "undefined" ) {
if ($(window).scrollTop() > headerHeight){
$("#mvp-main-nav-small").addClass("mvp-nav-small-fixed");
$("#mvp-main-body-wrap").css("margin-top", navHeight );
} else {
$("#mvp-main-nav-small").removeClass("mvp-nav-small-fixed");
$("#mvp-main-body-wrap").css("margin-top","0");
}
if ($(window).scrollTop() > headerHeight + 50){
$("#mvp-main-nav-small").addClass("mvp-fixed");
$("#mvp-main-nav-small").addClass("mvp-fixed-shadow");
$(".mvp-fly-top").addClass("mvp-to-top");
if(scroll < previousScroll) {
$("#mvp-main-nav-small").removeClass("mvp-soc-mob-up");
$("#mvp-soc-mob-wrap").removeClass("mvp-soc-mob-up");
$(".mvp-drop-nav-title").removeClass("mvp-nav-small-post");
$(".mvp-nav-menu").show();
} else {
$("#mvp-main-nav-small").addClass("mvp-soc-mob-up");
$("#mvp-soc-mob-wrap").addClass("mvp-soc-mob-up");
$(".mvp-drop-nav-title").addClass("mvp-nav-small-post");
$(".mvp-nav-menu").hide();
}
} else {
$("#mvp-main-nav-small").removeClass("mvp-fixed");
$("#mvp-main-nav-small").removeClass("mvp-fixed-shadow");
$(".mvp-fly-top").removeClass("mvp-to-top");
}
} else {
if ($(window).scrollTop() > navHeight){
$("#mvp-main-nav-small").addClass("mvp-nav-small-fixed");
$("#mvp-main-body-wrap").css("margin-top", navHeight );
} else {
$("#mvp-main-nav-small").removeClass("mvp-nav-small-fixed");
$("#mvp-main-body-wrap").css("margin-top","0");
}
if ($(window).scrollTop() > navHeight + 50){
$("#mvp-main-nav-small").addClass("mvp-fixed");
$("#mvp-main-nav-small").addClass("mvp-fixed-shadow");
$(".mvp-fly-top").addClass("mvp-to-top");
if(scroll < previousScroll) {
$("#mvp-main-nav-small").removeClass("mvp-soc-mob-up");
$("#mvp-soc-mob-wrap").removeClass("mvp-soc-mob-up");
$(".mvp-drop-nav-title").removeClass("mvp-nav-small-post");
$(".mvp-nav-menu").show();
} else {
$("#mvp-main-nav-small").addClass("mvp-soc-mob-up");
$("#mvp-soc-mob-wrap").addClass("mvp-soc-mob-up");
$(".mvp-drop-nav-title").addClass("mvp-nav-small-post");
$(".mvp-nav-menu").hide();
}
} else {
$("#mvp-main-nav-small").removeClass("mvp-fixed");
$("#mvp-main-nav-small").removeClass("mvp-fixed-shadow");
$(".mvp-fly-top").removeClass("mvp-to-top");
}
}
previousScroll = scroll;
});
});jQuery(document).ready(function($) {
// Video Post Scroll
$(window).on("scroll.video", function(event){
var scrollTop = $(window).scrollTop();
var elementOffset = $("#mvp-content-wrap").offset().top;
var distance = (elementOffset - scrollTop);
var aboveHeight = $("#mvp-video-embed-wrap").outerHeight();
if ($(window).scrollTop() > distance + aboveHeight + screen.height){
$("#mvp-video-embed-cont").addClass("mvp-vid-fixed");
$("#mvp-video-embed-wrap").addClass("mvp-vid-height");
$(".mvp-video-close").show();
} else {
$("#mvp-video-embed-cont").removeClass("mvp-vid-fixed");
$("#mvp-video-embed-wrap").removeClass("mvp-vid-height");
$(".mvp-video-close").hide();
}
});$(".mvp-video-close").on("click", function(){
$("#mvp-video-embed-cont").removeClass("mvp-vid-fixed");
$("#mvp-video-embed-wrap").removeClass("mvp-vid-height");
$(".mvp-video-close").hide();
$(window).off("scroll.video");
});});jQuery(document).ready(function($) {
// Mobile Social Buttons More
$(".mvp-soc-mob-right").on("click", function(){
$("#mvp-soc-mob-wrap").toggleClass("mvp-soc-mob-more");
});
});jQuery(document).ready(function($) {
$(".menu-item-has-children a").click(function(event){
event.stopPropagation();});$(".menu-item-has-children").click(function(){
$(this).addClass("toggled");
if($(".menu-item-has-children").hasClass("toggled"))
{
$(this).children("ul").toggle();
$(".mvp-fly-nav-menu").getNiceScroll().resize();
}
$(this).toggleClass("tog-minus");
return false;
});// Main Menu Scroll
$(".mvp-fly-nav-menu").niceScroll({cursorcolor:"#888",cursorwidth: 7,cursorborder: 0,zindex:999999});
});
https://www.uniquenewsonline.com/wp-content/themes/zox-news/js/scripts.js
https://www.uniquenewsonline.com/wp-content/themes/zox-news/js/retina.js
https://www.uniquenewsonline.com/wp-content/themes/zox-news/js/flexslider.js
https://www.uniquenewsonline.com/wp-includes/js/comment-reply.min.js
(function(w,d, s, id) {w.webpushr=w.webpushr||function(){(w.webpushr.q=w.webpushr.q||[]).push(arguments)};var js, fjs = d.getElementsByTagName(s)[0];js = d.createElement(s); js.async=1; js.id = id;js.src = "https://cdn.webpushr.com/app.min.js";
d.body.appendChild(js);}(window,document, 'script', 'webpushr-jssdk'));
webpushr('setup',{'key':'BPjbvHzuEYRqFV4yiEu3fIxgQ_hu6jxsQjNdj6baiXPpbFathx1pQk1KL7M1lXEoliGJckK0dG6UeYUZuTIiCRQ','sw':'https://www.uniquenewsonline.com/wp-content/plugins/webpushr-web-push-notifications/sdk_files/webpushr-sw.js.php'});
window.lazyLoadOptions = {
elements_selector: "iframe[data-lazy-src]",
data_src: "lazy-src",
data_srcset: "lazy-srcset",
data_sizes: "lazy-sizes",
class_loading: "lazyloading",
class_loaded: "lazyloaded",
threshold: 300,
callback_loaded: function(element) {
if ( element.tagName === "IFRAME" && element.dataset.rocketLazyload == "fitvidscompatible" ) {
if (element.classList.contains("lazyloaded") ) {
if (typeof window.jQuery != "undefined") {
if (jQuery.fn.fitVids) {
jQuery(element).parent().fitVids();
}
}
}
}
}};
window.addEventListener('LazyLoad::Initialized', function (e) {
var lazyLoadInstance = e.detail.instance;if (window.MutationObserver) {
var observer = new MutationObserver(function(mutations) {
var image_count = 0;
var iframe_count = 0;
var rocketlazy_count = 0;mutations.forEach(function(mutation) {
for (i = 0; i < mutation.addedNodes.length; i++) {
if (typeof mutation.addedNodes[i].getElementsByTagName !== 'function') {
return;
}if (typeof mutation.addedNodes[i].getElementsByClassName !== 'function') {
return;
}images = mutation.addedNodes[i].getElementsByTagName('img');
is_image = mutation.addedNodes[i].tagName == "IMG";
iframes = mutation.addedNodes[i].getElementsByTagName('iframe');
is_iframe = mutation.addedNodes[i].tagName == "IFRAME";
rocket_lazy = mutation.addedNodes[i].getElementsByClassName('rocket-lazyload');image_count += images.length;
iframe_count += iframes.length;
rocketlazy_count += rocket_lazy.length;if(is_image){
image_count += 1;
}if(is_iframe){
iframe_count += 1;
}
}
} );if(image_count > 0 || iframe_count > 0 || rocketlazy_count > 0){
lazyLoadInstance.update();
}
} );var b = document.getElementsByTagName("body")[0];
var config = { childList: true, subtree: true };observer.observe(b, config);
}
}, false);
https://www.uniquenewsonline.com/wp-content/plugins/rocket-lazy-load/assets/js/16.1/lazyload.min.js











 Photos1 week ago
Photos1 week ago
 Photos1 week ago
Photos1 week ago
 Entertainment5 hours ago
Entertainment5 hours ago
 Entertainment3 days ago
Entertainment3 days ago
 Web Series6 days ago
Web Series6 days ago
 Photos1 week ago
Photos1 week ago
 Web Series4 hours ago
Web Series4 hours ago
 Web Series4 days ago
Web Series4 days ago













